|
The array is a collection of elements (variables) that can be identified by at least one array, which is a key or index. This gives ice a lower density than water, which is why ice floats on water. Since there are fewer molecules in a given space, the water molecules are more spread out. This means that there are fewer water molecules in a given space than there are ice molecules. The molecules of ice are also shaped like small spheres, but they are slightly larger. Water is made up of molecules that are shaped like small spheres. Why Does Ice Float On Water BrainlyĬredit: The scientific explanation for why ice floats on water has to do with the molecules that make up water and ice. Ice cubes float because they are less dense than water. Because the density of salt water does not allow ice to float, it is difficult to keep ice submerged in salt water. Because ice floats on water, it is commonly used to determine the depth of the water. It is natural for lakes and rivers to freeze from top to bottom, allowing fish to survive even after the surface has been frozen.īecause ice is denser, it floats more easily on water and has a smooth surface. 
Water is made up of two types of positively charged hydrogens and two types of negatively charged oxygens. A solids has a higher density than a liquid. 
According to Ice, water is 9% denser than ice. This concept is explained in an ABC Science segment by putting a bucket of water and some rocks together. When a float moves, the weight of fluid displaced by the object decreases to the same weight as it is at rest. When a mixture has fewer objects in it than the other components (e.g., less dense), it floats. The density of an object determines whether or not it can sink. 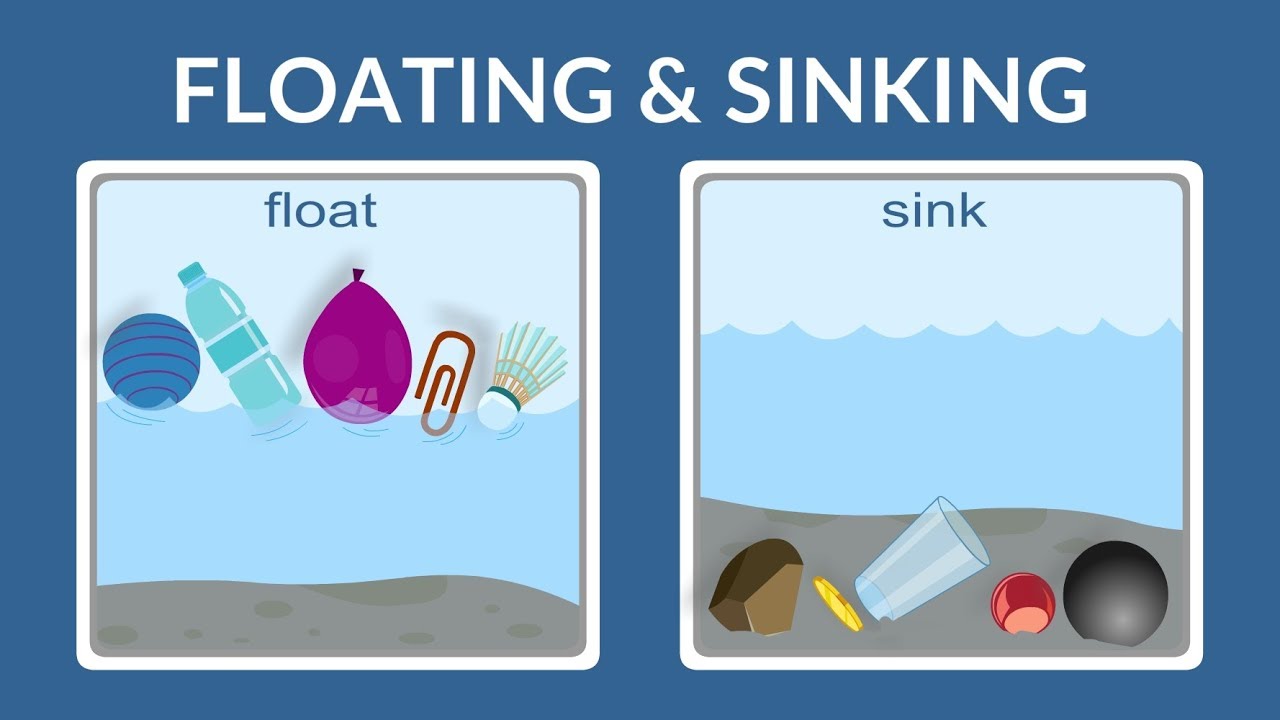
In other words, when you put ice cubes in water, they float on the surface. Why Does The Ice Cube Float And The Water Sink?Īccording to this, ice has a lower density (is less compact) than liquid water because the same mass of both spreads out and takes up more space when frozen. As a result, objects with a smaller density float in liquid.īecause ice is lighter than water, it floats in liquid form. As a result, the upthrust of the ice is greater than that of the ice. When there is less density, an object floats on top of the liquid it is in.Īccording to a verified expert, ice has a density that is lower than that of water. The simplest way to determine whether ice is less dense than water is to measure its density. Sinking and floating are the primary distinctions between the terms, with floating being the most important. It is possible for fish to survive in the frozen water because ice floats on the surface. Ice is a rigid crystal structure formed by the breakdown of negatively charged oxygen atoms in the presence of weaker hydrogen bonds as water temperature decreases. There are six hydrogen atoms in water molecules, each with a strong bond between them and one oxygen atom. In a density of 40o F, there is no limit to the amount of water in the body. solids are generally more dense than liquid substances. 
Why do ice cubes float in water? In contrast to water, ice floats because it is less dense. Tianna has been named today’s Wonder of the Day. And, as anyone who has tried to make a cocktail on a hot day knows, it is this property of ice that makes it so useful for keeping drinks cold. As a result, the ice floats on the water. This expansion of the water molecules creates pockets of air in the ice, and it is these pockets of air that make the ice less dense than the water. This is because the water molecules are able to move further apart from each other in the solid state than they can in the liquid state. But why is that? The answer has to do with the fact that water is less dense in its solid state than in its liquid state. As anyone who has ever made a cocktail knows, ice cubes float in water.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
